Keywords
Morgellons, Lyme disease, filaments, delusional parasitosis, dermatitis
Morgellons, Lyme disease, filaments, delusional parasitosis, dermatitis
There has been considerable debate about the disorder referred to as Morgellons disease (MD), which has no general medical acceptance as a disease entity. Rather it has been labeled ‘delusions of parasitosis’ or ‘dermatitis artefacta’ with enormous stigma attached to these terms and patients have been inappropriately medicated as discussed later. The dermopathy is characterized notably by filaments found under or protruding from the skin on microscopic examination from any area of anatomy without predilection for any particular site. Analysis has shown these to be monofilamentous exhibiting multiple colors including black, red, orange, blue, purple, white and clear1,2. They also fluoresce under UV light (except for the black ones)1,2. In 2011 similarity between MD and bovine digital dermatitis (BDD), a known spirochetal disease of cattle, was identified and published3. In 2013 Morgellons tissue was demonstrated to contain keratin and collagen filaments by several staining methods4. Spirochetal loads were found by light microscopy at 1000x magnification with silver nitrate staining and also by immunofluorescent staining with polyclonal anti-borrelial antibodies at 400x and 1000x magnification4,5. Further analysis using real time PCR demonstrated that the spirochetes were borrelial species5. This article firstly analyses the position of these findings in dermatology with comparative and differential analysis at the dermal level but then also highlights the characteristics that distinguish the dermopathy as part of a multisystem disease that principally involves arthritic, cardiac, neurological and psychological or psychiatric components. Further it is proposed that the diagnosis of delusions of parasitosis should be replaced with a novel described dermopathy as a feature of a multisytem disease.
A PubMed search using the term "Morgellons" on the 28th January 2013 returned 42 articles. From a historical perspective, after filtering these results for an organic rather than delusional or psychiatric causation the following milestones are noted. The term Morgellons was reported by Kellett in 1935 as being coined in 1674 by Sir Thomas Browne in his monograph entitled "De vermiculis capillaribus infantium"6. The affected child described experienced critical break outs of hair-like extrusions from the back, which upon occurring relieved the child from "coughs and convulsions". This information was re-reported and further elucidated historically by Emslie-Smith in the British Medical Journal in 19467. The terminology was reapplied around 2002 to describe the phenomenon of fine filaments found in skin using at least 60x magnification1. Mary Leaitao had found the terminology while researching her son’s dermopathy thus popularizing the term in a modern context. In 2010 Savely and Stricker described a population with microscopically confirmed filaments2. In 2011 Middelveen and Stricker published a novel perspective demonstrating marked similarity between MD and BDD3. The latter affects the skin above the hooves of cattle causing sores and lameness with associated economic loss and has been known and accepted as a spirochetal disease in the veterinary profession since 19748.
In 2012 Pearson et al. published a long awaited Centre of Disease Control (CDC) sponsored study on the possible causes of MD9. The key findings of this study of only 115 patients were: median age 52, 77% female, 77% Caucasian, 70% chronic fatigue symptoms, 54% reported poor state of health, 59% were found to have cognitive defects on testing, 63% had somatic complaints, 50% had detectable drugs in hair analysis and 78% reported exposure to solvents. Psychological issues spanned poor attention and memory, highly significant somatic disorders, depression and 24% had significant alcohol and drug use. Histological examination showed 57% solar elastosis or otherwise features consistent with arthropod bites or chronic excoriation. No parasites or mycobacterium were detected. Finally materials harvested were cotton or silicon. In the last paragraph of that paper it states under the discussion:
"We were not able to conclude based on this study whether this unexplained dermopathy represents a new condition, as has been proposed by those who use the term Morgellons, or wider recognition of an existing condition such as delusional infestation, with which it shares a number of clinical and epidemiologic features".
However in view of the prevalence of psychological issues found in the study the authors make the final recommendation:
"In the absence of an established cause or treatment, patients with this unexplained dermopathy may benefit from receipt of standard therapies for co-existing medical conditions and/or those recommended for similar conditions such delusions infestation".
One can interpret an open finding from this study, though the difficulty of showing an absence of an organic cause is acknowledged. In the study, matters pertaining to skin biopsy, coexisting multisystem disease and psychiatric co-morbid conditions are presented in a manner that aligns with the content of this review, as will be discussed. Also, it is noteworthy that this study used symptomatology exceeding three months duration as an exclusion criterion, which is not presented until the discussion on page nineteen, paragraph two of the article. This would have impacted markedly on incidence/prevalence figures and also the detection of multisystem disease in longer standing cases.
In contrast most of the results from the PubMed search mentioned at the beginning of this section, are dedicated to the diagnosis ‘delusions of parasitosis’. A further search using the term "delusions parasitosis" on the same date returns 320 findings all of which present non-organic illnessness. It is not possible to find a single paper amongst these with substantiated psychiatrically diagnosed delusional disorder in line with the discussion in the ‘Clinical features–psychiatric’section of this article.
Savely and Stricker found an association between MD and tick-borne diseases and hypothyroidism, a high prevalence of the condition in middle-aged white women and an increased prevalence of both smoking and substance abuse2. Although depression was noted in MD a pre-existing delusional disease was not reported. A retrospective study on delusions of parasitosis by Foster et al. analyzing Mayo Clinic, Rochester, Minnesota records from 2000 to 2007 gives a valuable insight as to whether these people in fact had MD, and shows a 75% female prevalence10. Symptom duration averaged 2.3 years with a range of 2 weeks to 23 years and more than 80% of patients were older than 50 years10.
The symptoms described in this section are those noted by the first and last authors in their clinical practice. It is fair to say that much of the citable literature, as referred to above, describes this same symptomatology.
Patients present with severe itching, pain, crawling or stinging sensations which may be localized, and may describe symptoms similar to formication or something growing in and extruding from the skin. There may be no visual dermopathy in early presentation. Patients may appear obsessed or even distressed with their symptoms and may describe finding filaments or tiny dark specks (some of which glitter) on or in the skin (Figure 1). Often patients have used magnification devices and may present with a container that demonstrates the offending particle or bug, which is usually dismissed as eschar debris by the professional and has been termed the ‘matchbox sign’11. Sometimes there is a belief that a bug or mite is growing in the skin and some will report that they have seen them. Questioned on this the patient may state they see little insects that fly past their eyes. It is plausible that these insects are attracted to the detritus of the damaged surface. In other cases several people (particularly familial, of which a case is briefly discussed hereunder) may develop the disorder at the same time and in this scenario the patient often believes they have transmitted the condition to family or friends. Sometimes patients exhibit highly compulsive behavior with excessive cleaning of the home in the fixed belief that the environment is infective. Temporary marital separation for the good health of the partner is not uncommon.
In these scenarios it becomes important to the patient to find evidence of filament disease, if they have not already done so, by magnification of itchy skin or evident dermopathy. Examination with a high power magnification of at least 60x is essential in the search for the presence of filaments under unbroken skin1–3. It is important not to fall into the trap of mistaking cotton and synthetic filaments for Morgellons filaments (the latter are typically smaller) and anyone familiar with a dermoscope knows all too well that the former are frequently seen loose on the skin or adhered to a damaged or ulcerated surface. Figure 2 shows cotton fibers adhered to sebaceous hyperplasia on the face of a 65 year old man using dermoscopy as well as a dermoscopic comparison between a MD wound ball filament and an overlying piece of cotton. Note that the single fiber strands on the side of the twisted cotton thread are closer in size to that of the MD fiber depicted here. The fact that the CDC study only used a dermoscope in the search for filaments may explain why only cotton fibers were found in that study, and so the opportunity to find the smaller filaments found in MD was missed9.
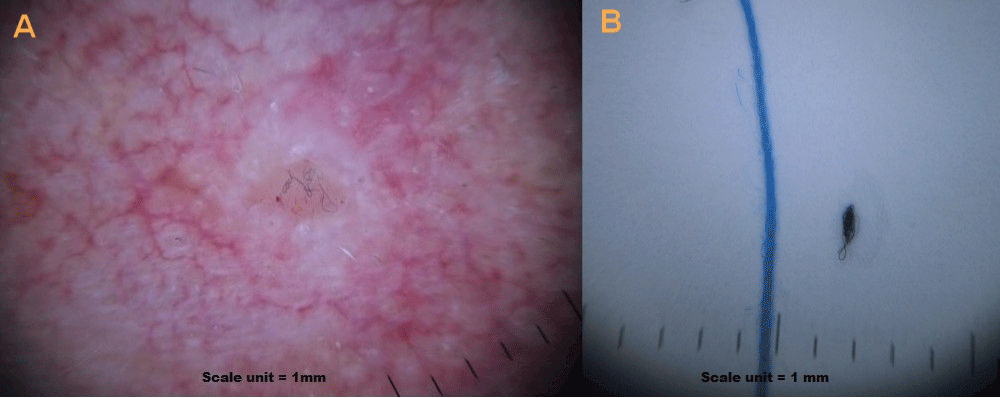
A) Dermoscopy view of cotton fibers adhered to sebaceous hyperplasia. B) Dermoscopic comparison to a Morgellons wound ball filament with an overlying piece of cotton (blue).
In more advanced forms of the disease there are widespread chronic elevated lesions, sometimes papular but more usually a plaque, ranging in size from 5 mm to 2 cm or more, and generally with recurrent surface eschar. Rarely the plaques can be huge (up to the length of the thigh). There may be ulcers and considerable eschars at more advanced stages.
If the disorder was not chronic, it could easily be mistaken for impetigo or other infective presentations such as staph folliculitis. Persistence for many weeks raises the differential diagnosis of scabies but this can be ruled out if careful observation shows that every wound is persistent and cycling over a 3–4 week period, and no mites or track marks are found.
Nail dystrophy may be present, somewhat similar to a psoriatic rather than fungal appearance, though the distinction can at times be difficult. Mucosal surfaces may be involved12,13. Patients have also described sudden deterioration of dental health. This is so far unreported in the literature. Of note is that treponema are considered normal oral flora but a recent study by Dabu et al. implicates a treponemal role in periodontal disease14.
The condition may also be compared to the dermal manifestation of syphilis in its secondary and tertiary stages where pruritus can be a dominant feature, and mucous membrane as well as nail dystrophy changes may be present15–20. It is also important to distinguish MD from intravenous drug abuse where formication can be a feature.
Lichenification may be present from the continual scratching and distinguishing MD from eczema by the detection of MD filaments is thus important. Post inflammatory hyper and hypo-pigmentation, depending on Fitzpatrick skin type, is a long standing feature of MD (Figure 3). There may be changes of lichen simplex chronicus in an older lesion and eventually ulceration. A widespread nodular appearance could be mistaken for nodular prurigo (Figure 4). There can be widespread pruritus without skin inflammation or restricted areas of inflammation.
The dermal presentation of MD is therefore a confusing picture though it appears that the inflammation and/or lesions are caused by dermal mechanical stress and irritation. It is proposed the mechanical stress is both internal and external from the filaments and scratching respectively. It is very important to note that the patient will report that they are scratching and damaging the skin surface, unlike the denial that is known to occur in factitial dermatitis21.
The authors propose that MD is a manifestation of Lyme disease (LD) which is otherwise called borreliosis, an infection vectored to humans by ticks. This section will therefore focus on LD, as will other sections in this manuscript.
Along with the previously described dermatological presentation it is necessary for the practitioner to consider the presence of symptoms or signs of multisystem disease. Attention must be given to evidence of arthritic (large joint mono or pauci arthritis), cardiac (AV blocks, carditis, pericarditis and pancarditis) and neurologic disorder22–24. In a recent review Biesieda has covered the scope of these presentations25. Gastrointestinal, genitourinary, gestational, musculoskeletal, nephritic and ophthalmic problems may occur26–29. Symptomatic history can extend for years. Pediatric LD has extensive similarities to adult forms of the condition (except for meningopolyradiculoneuritis and acrodermatitis chronica atrophicans, which are typically not seen in children) as noted in a comparative review by Esposito and co-authors30. A very comprehensive set of guidelines for LD diagnosis and management (2010) is available from Deutsche Borreliose-Gesellschaft, the German Borreliose Society and adequately sets out the spectrum of the disease and its management. It is available at http://www.borreliose-gesellschaft.de/Texte/guidelines.pdf. Dermatologists are well versed in the knowledge of conditions requiring urgent intravenous antibiotic therapy. In a 2012 review Sambrano and co-authors set out recent recommendations on therapy and dosages for a range of diseases with dosages including LD with its variant presentations and the necessities of oral versus intra venous therapy31.
Appropriate referral may also be considered as the dermatologist may restrict his interest in the patient to dermatological symptoms. In the past the dermatologist in a MD scenario has been diagnosing a psychiatric disorder and furthermore offering and suggesting psychotropics in a manner beyond and outside his sphere of expertise as discussed in the next section.
In addition to the dermatological, constitutional and systemic symptoms described, there are usually components of psychiatric morbidity in patients with MD. The first, third and last authors have encountered a range of illness across the depressive/anxiety spectrum in their clinical encounters with MD. Unfortunately, non-psychiatric medical practitioners or un-informed psychiatrists often label these patients as delusional, specifically with a well circumscribed somatic delusion which includes belief in infestation with insects or imagined inanimate objects such as filaments and particles. While these patients do suffer from psychiatric morbidity it is most often in the depressive and anxiety disorders spectrum and not likely a primary psychotic process.
A confounding factor may be that the pathophysiology of MD itself somehow impacts the peripheral or central nervous system in ways that create psychiatric symptoms (disorientation, visual changes or hallucinations) or amplifies other stimuli (tactile etc.) and results in a misperception of a real stimulus to be perceived as something else altogether. Of note, the case of misperception of a real stimulus does not qualify as a psychotic symptom as in fact the symptom is based in reality, or on real stimuli. According to the Diagnostic and Statistical Manual of Mental Disorders fourth text revision (DSM-IV) TR32, delusional disorder is an illness characterized by the presence of non-bizarre delusions in the absence of other mood or psychotic symptoms. Delusions are false beliefs based on incorrect inference about external reality that persist despite the evidence to the contrary. These beliefs are not ordinarily accepted by other members of the person's culture. The subtypes of delusion in the DSM-IV TR include: erotomanic, grandiose, jealous, persecutory, somatic, mixed, and unspecified.
Interestingly, patients with MD are diagnosed with delusions of parasitosis or infestation, neither of which exists as a clinical entity in the American classification of psychiatric disorder. Neither do these diagnoses exist in the World Health Organisation International Classification of Diseases33 where the closest diagnosis would be factitious dermatitis. From a psychiatric viewpoint the closest diagnostic category in the DSM-IV TR would be the subtype of somatic delusions, referring to the belief held by an individual that they have a physical defect or general medical condition that has yet to be identified. The medical profession must be careful when making a diagnosis of somatic delusion for certain specific reasons. First, history has proven that the medical establishment has often misconceived a collection of symptoms, or syndrome, to be psychological in origin (or psychologically mediated) rather than physiological. A prominent example is the aetiology of Peptic Ulcer Disease, once believed to be stress induced but now known to be caused by helicobacter infection34. The consequences of such misdiagnosis include delay in appropriate treatment, worsening of the actual disease process, and punitive labelling of the patient as "psychiatric", which in itself can have several adverse outcomes such as psychtropic medication side effects.
Second, the diagnostic criteria for delusional disorder, as outlined below, are quite specific in guiding clinicians (specifically mental health professionals) on how to arrive at this "rule out" or diagnosis of exclusion. The most recent criteria for a delusional disease, which are not manifest in MD patients, include:
A: Nonbizarre delusions occurring for at least 1 month's duration involving situations that occur in real life, such as being followed, poisoned, infected, loved at distance, deceived by spouse or lover, or having a disease.
B: DSM-IV-TR Criteria A for schizophrenia has never been met. Patients do not have simultaneous hallucinations, disorganized speech, negative symptoms such as affective flattening, or grossly disorganized behavior. Note: Tactile and olfactory hallucinations may be present in delusional disorder if they are related to the delusional theme.
C: Apart from the impact of the delusion(s) or its ramifications, functioning is not markedly impaired and behavior is not obviously odd or bizarre.
D: If mood episodes have occurred concurrently with delusions, their total duration has been brief relative to the duration of the delusional periods.
E: The disturbance is not due to the direct physiological effects of a substance, either a drug of abuse or a medication, or a general medical condition.
The "matchbox sign" and "meme" are oft quoted reasons supporting a diagnosis of psychotic beliefs in patients presenting with symptoms of MD. The "matchbox sign" is the term applied to the scenario where a patient brings in samples of what they believe to be foreign material from their own bodies to their physician for inspection. This behavior was first described by Perrin in 1896 but the term "matchbox sign" was not coined until a 1983 case presentation in The Lancet in which the author noted the use of the matchbox as a device to store and present a specimen11. The "meme" is the concept of an idea or behavior that spreads from person to person within a culture; it acts as a unit for carrying cultural ideas which can be transmitted from one mind to another through writing, speech, rituals or other forms (i.e. internet) of communication. In 2009 the paper "Morgellons Disease as Internet Meme" was published in the psychiatric journal Psychosomatics, further spreading the belief that MD is a fabrication of man’s psyche, a fabrication that can be transmitted to others as easily as its symptoms are reported on various websites35.
Interestingly, various fields of medicine hold the randomized, controlled, double blinded study to be the gold standard by which to measure efficacy or statistical associations in diagnosis, treatment, and prognosis. The authors are unaware of any studies that use a standardized and methodological approach to prove the relationship between bringing samples of perceived infesting specimens to a physician and the appropriate diagnosis of delusional disorder. In the same vein they are also unaware of studies that can demonstrate that access to the internet or to knowledge obtained on the internet about MD acts as the inciting factor or as a propagating vector for the spread of a delusional disorder. If this were the case, and given the explosion of media forms (radio, television, internet, mobile devices etc.), one might expect a far more drastic rise in the reported cases of delusional disorders of all types over the past 100 years.
An appropriate comparison grouping would be arthropod bites, dermatitis artefacta, drug reaction, eczema including nummular and discoid types, impetigo, lichen planus, lichen simplex chronicus, nodular prurigo, scabies including Norwegian scabies, tinea corporis, toxoplasmosis (cutaneous) and severe pruritus from systemic disease e.g. lymphoma and syphilis (secondary and tertiary). The tropical diseases cutaneous larva migrans, leishmaniasis, gnathostomiasis and schistosomiasis may have to be considered if there is a positive travel history but these should be specifically diagnosable. The perforating dermatoses have been described and compared to MD by Savely, Leitao and Stricker1. These include acquired perforating dermatosis, elastosis perforans serpiginosa, Kyrle disease, perforating folliculitis and reactive perforating collagenosis. The banishment of the concept of delusions of parasitosis as accepted medical terminology is proposed and it should be noted that there is no such listing as a recognised entity in the DSM-IV (TR) or the upcoming DSM-5 as discussed above.
MD has been defined as the presence of varying coloured filaments visible with light microscopy at a magnification of at least 60x in or protruding from skin lesions or under unbroken skin2. Detecting the presence of spirochetes may be possible by light microscopy, but now the detection of borrelial species by culture, staining, immunofluorescent staining, electron microscopy or PCR is an equally valid proof in the presence of MD symptoms5. However there is also the aspect of describing the character of the dermis as affected by these filaments, and this is discussed below showing a multivariate presentation and histology confirming dermatitis. The presence of itch and inflammation makes this condition a dermatitis as distinct from a dermatosis, though as discussed, pruritus can be a leading symptom with no skin change evident. The reverse scenario of a patient with borrelial infection describing a coexistent skin condition with a similar time line requires close consideration of the possibility of MD. Also of note is the fact that erythema migrans (EM) lesions may be described by patients as itchy and painful, and generalized pruritus has been noted in some patients with EM, the implication being that MD can have an instantaneous onset, which is further discussed in the next section36. Finally, typical features of MD involve persistence, recurrence, varying sites and cyclical dermal involvement.
Often two or more members of a family can be affected simultaneously. The lead author can report multiple instances in his practice; however in one scenario a family of three were infested with ticks after an Indian myna bird, Acridotheres tristis, was trapped in their house on the east coast of Australia for two days while they were away. The young wife and twelve month old child proceeded to develop symptomatology of MD. At first clinical presentation to the lead author six months later, the wife presented symptoms of MD together with some evidence of LD, reporting brain fog, poor concentration and memory, dental pain, depression, anxiety, panic, insomnia, irritability and right sided facial pain in the distribution of the second and third division trigeminal nerve. All symptoms were new and commenced with the MD. No neurological deficit was demonstrable. A diagnosis of Morgellons and neurological Lyme was made, confirmed with positive real-time PCR for Borrelia sp. on whole blood at Australian Biologics in Sydney, Australia. The method is as previously described5 and is also presented below. Blood was also forwarded to IGenex California USA for western blot analysis which was positive for LD. IgM was evident at bands 18+, 31+, 41+ and 58++ kDa. The IgG showed indeterminate bands at 31, 34 and 39 kDa and positive at bands 41++, 45+ and 58+ kDa. She presented with photos of a fed nymph tick removed from her body (which at the time was not formally analysed) and also a black speck from her skin for my analysis. She had examined the speck under 200x magnification and found a black ball of twisted filament. That speck is presented at 500x magnification showing a ball of firmly wound filament which was red at the higher magnification (Figure 5).
Dermoscopic scanning for anything smaller than a textile filament may be helpful but it is very difficult at this 10x resolution to identify Morgellons filaments with certainty. Higher magnification is required. Savely and Stricker recommended a minimum of 60x2. An eschar specimen from a female Morgellons patient in her late forties was obtained and bisected. The dermoscopic view is shown in this case and does show filaments at 10x magnification (Figure 6). See Figure 7 for a light microscopy image of the same material at 500x. See Figure 8 for Scanning Electron Microscopy (SEM) images of the same material (note the frond like appearance).


In a different male patient in his late forties with a shaved scalp, blue filaments appearing to arise from the infundibulum of a hair shaft were imaged (Figure 9). This image was obtained by direct observation of skin using a 500x digital microscope. It appears that the filaments are perpendicular to the hair and leaving the hair follicle at the level of the infundibulum. The smallest is approximately 2.5 microns in diameter. The filaments appear to originate from the outer sheath of the hair apparatus and the source could be anywhere from above the sebaceous gland on the acute angle side or on the obtuse side above the erector pilii muscle and then up to the superficial flattened areas of the epidermis. Clinically the patient also had neuroborreliosis for the same time interval as his skin disease. Please refer to the endpoint polymerase chain reaction (PCR) analysis of this patient’s whole blood in the Endpoint section.
Skin histology may show acanthosis and spongiosis particularly in an early lesion. Other descriptors found are psoriasiform hyperplasia, thickened granular layer, compact hyperkeratosis and parakeratosis thus giving a rather confusing picture. Sometimes the filament will be seen and reported as extraneous material. However protein-specific staining has shown the filaments may be composed of keratin or collagen4. Middleveen and Stricker drew an association between Morgellons and bovine digital dermatitis as previously discussed3. In 2006 Savely and Stricker drew an association between Morgellons and LD, also a spirochetal infection2. Middleveen et al. showed spirochetal evidence in Morgellons tissue by microscopy, culture, immunostaining, scanning and transmission electron microscopy and showed the presence of Borrelia species with PCR evidence5. In MD the filaments, as well as growing outwards and in the horizontal plane, can grow through the dermis and into subdermal tissues with papilla formation (these papillae encase helically coiled filaments). See the under surface of an eschar from a diagnosed MD patient (Figure 10). Finally in biopsy reports from the skin of patients believed to have delusions of parasitosis, Hylwa describes dermatitis features on histological examination in 61% of patients studied, including 33 cases of chronic dermatitis, 10 cases of subacute dermatitis and 6 cases of lichen simplex chronicus37.
Spirochetal presence in the skin of diagnosed Morgellons patients has previously been demonstrated, including PCR detection of borrelial pathogens5. In this segment PCR validation of systemic borrelial infection with diagnosed MD is addressed.
Borrelial real time PCR testing was carried out on the aforementioned female patient (concurrent case presentation) at Australian Biologics by the fourth author in August 2012. B. burgdorferi was detected using the Eco™ Real-Time PCR system with software version 3.0.16.0. The sample was analysed with positive and negative controls using primers and a probe for the borrelial 16S gene target. The primers were AB-Bor1 (proprietary to Australian Biologics). The thermal profile for the analysis involved incubation for 2 mins at 50°C, polymerase activation for 10 mins at 95°C then PCR cycling for 40 cycles of 10 secs at 95°C dropping to 60°C sustained for 45 secs. Subject material showed a positive quantitative cycle Cq result of 36.39. Please see amplification plot where there are also two positive controls to the top and 2 negative controls at the base and Cq table to the right (Figure 11).
Borrelial PCR testing (endpoint) was carried out on the aforesaid male patient (discussed in the skin microscopy section) at Australian Biologics by the fourth author in April 2011. The method is tabulated viz:
DNA was prepared by blood collection (7 ml) into 1 ACD tube and centrifuged at 1200 rpm for 10 minutes. The buffy coat layer was removed by taking a small amount of red blood cells and plasma with the leukocytes then 500 µl of buffy coat cells were pipetted into a sterile 1.5 ml Eppendorf tube and 500 µl of Qiagen AL buffer added. Qiagen was sourced from their Australian distributor in Chadstone, Victoria. DNA was extracted using the Qiagen QIAamp® DNA Blood Mini Kit, according to the manufacturer’s instructions, and eluted in 75 μl of Qiagen AE buffer. Then primers targeting the Borrelia burgdorferi rpoC gene were used. The rpoC gene was amplified using Forward Primer 5’-AAATGGCTAAAGTAAGCGGAATTGTAC-3’ and Reverse Primer 5’-CAGAAATTCTGTAAACTAATCCCACC-3’, as described by Adelson M. E. et al38. In a 25 µl reaction consisting of 12.5 µl of Promega’s GoTaq® DNA Polymerase (Promega, Australia) 7.5 pmoles of each primer, 4.5 µl H2O and template of 5 µl. Product size 231 bp. Cycling conditions were initial denaturation at 95º for 45s followed by a 10 cycle Touchdown of 15s@95º, 45s@65º–56º and 45s@72º then 50 cycles 15s@95º, 45s@55º, 45s@72º with final extension of 2mins@72º. PCR products were analysed by standard 2.5% agarose gel. Product of the PCR amplification was purified using the Marligen Rapid PCR Purification Kit (Marligen Biosciences, Australia) following manufacturer’s instructions. The product was eluted with 50 µl of warmed TE buffer (Promega, Australia) and forwarded to Australian Genome Research Facility (AGRF) in Sydney, Australia for sequencing.
Serum endpoint PCR gave a positive result for borrelial species. The sequence produced was 5’-CGGGGGGCCGAGGGATCGTTGAAAGGTAAAGGCTTATTATATTTTAGATGAGTATGGGGTTGAC-3’. The post edited sequence was 5’-TTATTATATTTTAGATGAGTATGGGGTTGAC-3’. This sequence was then submitted for Basic Local Alignment Search Tool (BLAST) inquiry as previously described39,40. The results show the top two findings to be Borrelia garinii. Significant findings are presented in Table 1 and the top two alignments with two matches in Figure 12.
| Description | Max score | Total score | Query cover | E value | Max identity | Accession |
|---|---|---|---|---|---|---|
| Borrelia garinii NMJW1, complete genome | 50.1 | 80.3 | 90% | 4.00E-04 | 100% | CP003866.1 |
| Borrelia garinii BgVir chromosome linear, complete sequence | 50.1 | 80.3 | 90% | 4.00E-04 | 100% | CP003151.1 |
| Borrelia bissettii DN127, complete genome | 50.1 | 50.1 | 80% | 4.00E-04 | 100% | CP002746.1 |
| Borrelia burgdorferi N40, complete genome | 50.1 | 50.1 | 80% | 4.00E-04 | 100% | CP002228.1 |
| Borrelia burgdorferi JD1, complete genome | 50.1 | 50.1 | 80% | 4.00E-04 | 100% | CP002312.1 |
| Borrelia burgdorferi ZS7, complete genome | 50.1 | 50.1 | 80% | 4.00E-04 | 100% | CP001205.1 |
| Borrelia garinii PBi, complete genome | 50.1 | 80.3 | 90% | 4.00E-04 | 100% | CP000013.1 |
| Borrelia burgdorferi B31, complete genome | 50.1 | 50.1 | 80% | 4.00E-04 | 100% | AE000783.1 |
| Borrelia afzelii HLJ01, complete genome | 42.1 | 72.4 | 96% | 0.11 | 100% | CP003882.1 |
| Borrelia afzelii PKo, complete genome | 42.1 | 72.4 | 96% | 0.11 | 100% | CP002933.1 |
| Borrelia afzelii PKo, complete genome | 42.1 | 72.4 | 96% | 0.11 | 100% | CP000395.1 |
There has been considerable debate about MD treatments. To date, an exhaustive list of home remedies can be found on the internet, but evidence supporting the efficacy of these treatments is lacking. Segments of the profession have tried therapy with antihelminthics including ivermectin, mebendazole, praziquantel and albendazole, but this approach as sole therapy is by and large ineffective and false conclusions as to their efficacy can be drawn by the cycling nature of the dermopathy. No published material is available supporting such use. The lead author has found that adequate treatment of B. burgdorferi will suppress and control the disorder, more easily so in earlier disease (unpublished observations). Such treatment requires simultaneous antibiotic polypharmacy. Cell wall drugs are useful for the extracellular spirochete forms, tetracyclines or macrolides are useful for the intracellular forms, while imidazole compounds such as metronidazole and tinidazole are useful for the cystic forms of B. burgdorferi. In conjunction with LD treatment, the myriad associated coinfections need to be addressed including Anaplasma, Babesia, Bartonella, Ehrlichia and Rickettsia. With advancing knowledge of cell-wall deficient forms of the Lyme spirochete, the mechanisms associated with chronic protracted LD are being understood, albeit slowly41,42. This knowledge also helps us to understand why skin examination looking only for spirochetes can be unrewarding as given the right conditions both the intracellular and cystic forms can morph back to a spirochetal form. A recent study highlighted the significant burden of protracted LD patient care on healthcare systems43, and delayed diagnosis of MD and LD adds to that burden. We do not know if skin co-infections can be excluded at this juncture, nor can we rule out the possibility of co-factors playing a crucial role in filament formation. These are areas for ongoing research as is the exact mechanism of filament production. To date the long-term prognosis for protracted LD illness that includes MD remains unsettled.
LD or Lyme borreliosis as it is sometimes called, is borreliosis in the strictest sense. The dermatology profession now has to consider that in the same manner, the more correct terminology for MD would be ‘borrelial dermatitis’. ‘Spirochetal dermatitis’ could be considered as a suitable term but in the authors opinion is not specific enough as the pathogens appear to specifically belong to members of the genus Borrelia.
Borrelial dermatitis or MD is characterized by the progressive onset of skin irritation due to epidermal formation of filaments known to be keratin or collagen4 in nature and arising from the base of the epidermis and can arise from a disrupted hair follicle as demonstrated in Figure 9. Filaments are typically 5–50 microns in diameter. Extrusion through skin and entrapment under the skin leads to pruritus, formication, other tactile disturbances and subsequent skin damage, both internal and external, which at the extreme can manifest as large chronic ulcerations. The clinical appearance of skin can range from an eczema-like appearance through to that of nodular prurigo. Mucosal surfaces may similarly be affected and nail dystrophy may be present. Evidence of multi systemic LD (borreliosis) can be found. Spirochetal presence can be demonstrated and borrelial infection implicated5. Although it is not the authors’ belief that MD is primarily a psychotic disorder, it is evident that psychiatric comorbidity is often present in this patient group. Whether this is due to a pre-existing psychiatric diagnosis or one which develops such as acute stress reaction, adjustment disorder or post-traumatic stress disorder during the course of emerging MD symptoms and subsequent attempts at treatment, it is important to recognize psychiatric comorbidity. Timely identification and efficacious treatment of psychiatric symptoms can improve overall psychological well-being, possibly reduce the chance that patients avail themselves of dangerous and idiosyncratic treatments for MD symptoms, increase rapport and follow up with physicians in multiple specialities, and preserve the dignity and respect which is due to the patient. Hylwa and co-authors reported a significant load of psychiatric burden in a study of Mayo clinic patients with "delusional infestation" but psychiatric delusional conditions were not reported44. We argue that Morgellons disease, a disorder by and large dismissed by the profession as delusions of parasitosis, in particular by dermatologists and primary care physicians, and supposedly highly vectored by internet transmission should now be called borrelial dermatitis within the profession and acknowledged as a presentation of a multisystem infective disease (LD) and treated appropriately as such35. Research needs to be directed at identifying the fundamental flaw in filament production at the cellular level, which may involve RNA/DNA dysfunction, in the hope of addressing further treatment.
Written informed consent for publication of their clinical details and/or clinical images was obtained from the patients.
PM conceived the report and developed the dermatological aspects with JE. EK contributed all psychiatric components. JB carried out all PCR testing and with PM examined BLAST alignments. MM contributed to microbiology components. RS reviewed Morgellons features. PM and RS developed systemic LD presentation. PM prepared the first draft of the manuscript except for that component specifically made by EK. All authors were involved in the revision of the draft manuscript and have agreed to the final content.
PJM, EJK and RBS serve without compensation on the medical advisory panel of the Charles E. Holman Foundation. MJM serves without compensation on the scientific advisory panel of the Charles E. Holman Foundation. Each otherwise has no relevant competing interests to declare. JSE and JMB also have no conflicts to declare.
The lead author wishes to acknowledge the DPD course of the Department of Dermatology at Cardiff University and the DCPDS annual meeting in 2011 where he was given the opportunity to present a short talk on Morgellons which became the formative vessel for this paper in collaboration with the second author. The authors thank Dr Robert Allan for manuscript review.
| Views | Downloads | |
|---|---|---|
| F1000Research | - | - |
|
PubMed Central
Data from PMC are received and updated monthly.
|
- | - |
Competing Interests: No competing interests were disclosed.
Competing Interests: No competing interests were disclosed.
References
1. Abel IS, Marzagão G, Yoshinari NH, Schumaker TT: Borrelia-like spirochetes recovered from ticks and small mammals collected in the Atlantic Forest Reserve, Cotia County, State of São Paulo.Mem Inst Oswaldo Cruz. 2000; 95 (5): 621-624 PubMed Abstract | Free Full Text | Publisher Full TextCompeting Interests: No competing interests were disclosed.
Competing Interests: I have met Dr Peter Mayne, Dr. Ray Stricker and Dr Eva Sapi at international scientific meetings on Lyme disease [most recently at the ILADS International meeting Boston, MA, USA, Nov 2012]. I was first introduced to Dr Marianne Middelveen in November 2012. I have a position as Affiliated Researcher in the Dr Eva Sapi Research Borrelia Laboratory at the University of New Haven, as an expert in Borrelia morphology from my 35 years of research work in Lyme borreliosis. Dr. Ray Stricker has presented numerous publications and lectures on Lyme borreliosis at ILADS scientific meetings. Dr. Judith Miklossy has published extensively on the links between Neuroborreliosis-Chronic type and some cases of Alzheimer’s disease, and I have also separately published similar Lyme Chronic Neuroborreliosis/Alzheimer’s results. I have never attended any of the scientific meetings on the subject of Morgellons Disease. I have no personal or scientific biases either for or against the Morgellons entity. I can confirm that as a pathologist, that I have never received any compensation from any of the authors.
Alongside their report, reviewers assign a status to the article:
| Invited Reviewers | ||||
|---|---|---|---|---|
| 1 | 2 | 3 | 4 | |
|
Version 1 01 May 13 |
read | read | read | read |
Provide sufficient details of any financial or non-financial competing interests to enable users to assess whether your comments might lead a reasonable person to question your impartiality. Consider the following examples, but note that this is not an exhaustive list:
Sign up for content alerts and receive a weekly or monthly email with all newly published articles
Already registered? Sign in
The email address should be the one you originally registered with F1000.
You registered with F1000 via Google, so we cannot reset your password.
To sign in, please click here.
If you still need help with your Google account password, please click here.
You registered with F1000 via Facebook, so we cannot reset your password.
To sign in, please click here.
If you still need help with your Facebook account password, please click here.
If your email address is registered with us, we will email you instructions to reset your password.
If you think you should have received this email but it has not arrived, please check your spam filters and/or contact for further assistance.
Dr. MacDonald also commented that "Since my referee report was published the US Government has added Morgellons disease to the NIH office of Rare Diseases Research Genetic and Rare Diseases Information Center (GARD) list of rare diseases." I would point out that the NIH site that he lists simply points to the CDC report, the very same report I cited, which concluded that Morgellons is not a real disease: that there is "no infectious cause and no evidence of an environmental link." That report explains that the sores on the skin of patients self-diagnosed with Morgellons are due to "chronic scratching or picking, without an underlying cause." There is no evidence whatsoever that Morgellons is linked to Borrelia infection, and this article does not comprise such evidence.
Dr. MacDonald also commented that "Since my referee report was published the US Government has added Morgellons disease to the NIH office of Rare Diseases Research Genetic and Rare Diseases Information Center (GARD) list of rare diseases." I would point out that the NIH site that he lists simply points to the CDC report, the very same report I cited, which concluded that Morgellons is not a real disease: that there is "no infectious cause and no evidence of an environmental link." That report explains that the sores on the skin of patients self-diagnosed with Morgellons are due to "chronic scratching or picking, without an underlying cause." There is no evidence whatsoever that Morgellons is linked to Borrelia infection, and this article does not comprise such evidence.